AdvanTech Plastics provides ISO 13485–compliant medical device and pharmaceutical injection molding from controlled manufacturing environments, including ISO Class 7 and ISO Class 8 cleanrooms. We support regulated programs with documented quality systems, lot traceability, controlled handling procedures, and validation support for IQ/OQ/PQ protocols. From diagnostic components and drug-delivery parts to medical packaging and precision fluid-path components, our team delivers repeatable, compliant production built for today’s regulatory expectations.
With meticulous attention to all aspects of the medical injection molding process and a reputation for timely deliveries at globally competitive prices.
AdvanTech medical/dental injection molding is the optimal choice for efficient, cost-effective, FDA-compliant devices and components.
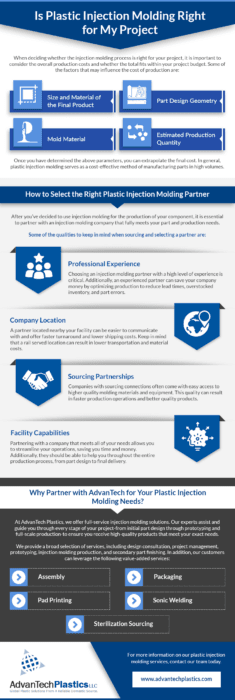
Regulated Medical & Pharmaceutical Capabilities
Precision injection molding for regulated markets:
* Multi-cavity high-volume production
* Insert molding & overmolding
* Micro / tight tolerance molding
* Automated part handling
* Assembly & packaging integration
ISO 13485 Quality System & Traceability
* Controlled SOPs
* Employee training records
* Calibration systems
* Lot traceability
* CAPA & nonconformance systems
* Documented production workflows
ISO Class 7 & 8 Cleanroom Manufacturing
AdvanTech Plastics operates ISO Class 7 and ISO Class 8 cleanroom environments for molding, assembly, and packaging of medical and pharmaceutical components. These controlled environments utilize HEPA filtration, defined gowning protocols, environmental monitoring, and documented cleaning procedures to reduce particulate contamination risk. Cleanroom operations are integrated into our ISO 13485 quality system and supported by controlled material flow, segregated production areas, and traceable lot documentation.
ISO Validation & Process Control (IQ / OQ / PQ Support)
We support medical device validation programs with documented process development and qualification activities. Our team assists with IQ (Installation Qualification), OQ (Operational Qualification), and PQ (Performance Qualification) protocol execution, along with process FMEA, control plans, gage R&R studies, and capability analysis (Cpk/Ppk) as required. Production parameters are documented and monitored to ensure repeatable, compliant manufacturing.
Applications We Support
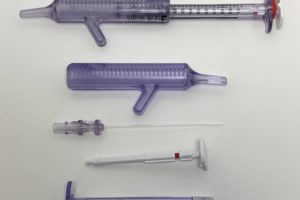
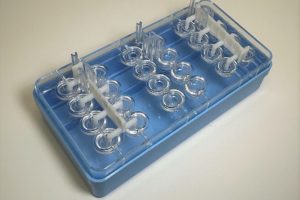
* Diagnostic cartridges & components
* Drug-delivery housings & caps
* Fluid-path components
* Medical packaging
* Laboratory disposables
* Precision enclosures
Request a Quote or Technical Review
Start Your Medical Device Program with Confidence
If you are evaluating a new medical device project, transferring existing tooling, or preparing for validation, our engineering team can review your design, material requirements, and regulatory expectations.
To begin, please provide:
3D CAD and 2D drawings with CTQs
Estimated annual volumes
Target resin and sterilization method
Cleanroom or packaging requirements
Validation scope (IQ/OQ/PQ if applicable)
Our team typically responds within one business day to schedule a technical discussion.